

Medical Devices
One of the biggest challenges facing healthcare practitioners during the peak of the COVID-19 pandemic is to help patients maintain timely and continued access to high-quality medical devices. Many hospitals and clinics had been forced to postpone or cancel elective medical procedures either out of safety concerns or because they need to focus resources on treating COVID-19 patients. These changes in the location and structure of care delivery, coupled with the advancement in biomedical technology, have given rise to new thinking about how medical devices can reach and provide services to healthcare providers and patients.
At Avalon, our main goal is to consistently provide superior experience to all users. Yet at the same time, our research teams are constantly responding to changes in the healthcare sector with innovations that seek to enhance the adaptability and flexibility of medical devices, meaning that they can be used in a variety of environments, including clinics, care homes or even domestic settings. Combining that with our commitment to quality and the vast demand for high quality products from the Greater Bay Area, Avalon is well-positioned to become the future leader in medical device for the region.

Up to 70% of newborns are affected by neonatal jaundice that causes the yellowing of skins and the white part of the eyes. If left untreated, it may lead to kernicterus, causing irreversible brain damage. The conditions are normally treated by phototherapy at hospitals, with the drawback of having to separate the infants from the parents from the first few days of birth. This is made even more complicated by the pandemic as many considered hospitals to be high-risk areas.
KangaLite, developed by Avalon Photonics, is a safe, effective, and most importantly portable phototherapy system that allows the treatment to be carried out at home. The lightweight device can be safely strapped onto the torso of an adult holding the infant, rendering maternal bonding without interrupting the treatment.
KangaLite is currently being used by several top-tier children hospitals in China, including Children’s Hospital of Chongqing Medical University and Tianjin Central Hospital of Gynaecology Obstetrics, as well as several private clinics in Hong Kong.
KangaLite is also in compliant with the following international guidelines and approvals:
Clinical practice has waited long for solutions that can provide quantitative liver function assessment for both management of liver diseases and for drugs that are metabolized by the liver. Our team at Avalon HepaPOC seeks to address this with the Galactocheck Blood Galactose Monitoring System, which contains a device that measures blood galactose and a galactose solution. It is intended to be used for qualitative measurement of blood galactose concentration, as a function of hepatic flow, and in fresh capillary whole blood samples drawn from the fingertips 60 minutes post-administration of galactose. This monitoring system is designed to be used in doctors’ offices as point-of-care units.
Clinical studies for the system have been completed in several key areas. It has recently been approved by the Taiwan FDA and is closing in on winning regulatory approval in the U.S. We look forward to bringing more good news about this exciting project!

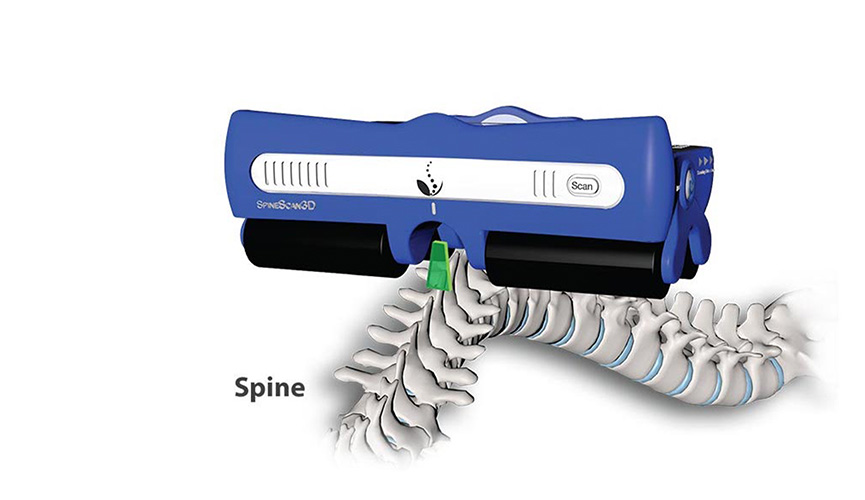
Scoliosis is widely-considered one of the most common spinal disorders, affecting up to 3% of adolescents worldwide. In major Chinese cities such as Beijing and Shanghai, the figure may exceed 15%, translating into 226 millions of adolescents in the country needing screening for the condition each year. Among those, around 11 million may need assessment and home monitoring.
To help meet this demand, our team at Avalon OrthoSmart and SpineCare have been working on a reliable and user-friendly system that can effectively improve spine health among the population. This one-stop solution will comprise of components such as AI-assisted screening, hardware and software for home monitoring, non-invasive assessment in specialist clinics, and treatment such as growth sparing surgeries.